Multiman ps3 games download games. Serial Dilution. This is commonly referred to as a 10x dilution. The illustration above follows the relationship between volume of solvent, number of molecules of solute and concentration of a solution over a set of 4 dilutions. The concentration can be tracked in M which is a common unit for chemistry, or particles per ml. Serial Dilution. This is commonly referred to as a 10x dilution. The illustration above follows the relationship between volume of solvent, number of molecules of solute and concentration of a solution over a set of 4 dilutions. The concentration can be tracked in M which is a common unit for chemistry, or particles per ml. Serial dilution. Serial dilution generally refers to selection preformed in the standard growth regimes typically used in the lab: flasks, test tubes, solid media, or 96-well plates. Cultures are usually allowed to grow through a normal growth curve, with daily transfer of a small volume of the expanded culture into fresh medium.
- What Is Serial Dilution Pdf
- What Is A Serial Dilution And Why Is The Technique Used
- How To Do Serial Dilutions
- What Is A 2 Fold Serial Dilution
- Serial Dilution Calculator Microbiology
Real-life chemists in real-life labs don’t make every solution from scratch. Instead, they make concentrated stock solutions and then make dilutions of those stocks as necessary for a given experiment.
To make a dilution, you simply add a small quantity of a concentrated stock solution to an amount of pure solvent. The resulting solution contains the amount of solute originally taken from the stock solution but disperses that solute throughout a greater volume. Therefore, the final concentration is lower; the final solution is less concentrated and more dilute.
How do you know how much of the stock solution to use and how much of the pure solvent to use? It depends on the concentration of the stock and on the concentration and volume of the final solution you want. You can answer these kinds of pressing questions by using the dilution equation, which relates concentration (C) and volume (V) between initial and final states:
C1V1 = C2V2
You can use the dilution equation with any units of concentration, provided you use the same units throughout the calculation. Because molarity is such a common way to express concentration, the dilution equation is sometimes expressed in the following way, where M1 and M2 refer to the initial and final molarity, respectively:
M1V1 = M2V2
For example, how would you prepare 500. mL of 0.200 M NaOH(aq) from a stock solution of 1.5 M NaOH?
Start by using the dilution equation,
What Is Serial Dilution Pdf
M1V1 = M2V2
The initial molarity, M1, comes from the stock solution and is therefore 1.5 M. The final molarity is the one you want in your final solution, which is 0.200 M. The final volume is the one you want for your final solution, 500. mL, which is equivalent to 0.500 L. Using these known values, you can calculate the initial volume, V1:
The calculated volume is equivalent to 67 mL. The final volume of the aqueous solution is to be 500 mL, and 67 mL of this volume comes from the stock solution. The remainder, 500 mL – 67 mL = 433 mL, comes from pure solvent (water, in this case). So to prepare the solution, add 67 mL of 1.5 M stock solution to 433 mL water. Mix and enjoy!
Try another problem: What is the final concentration in molarity of a solution prepared by diluting 2.50 mL of 3.00 M KCl(aq) up to 0.175 L final volume? Free ip address software.
You can use the dilution equation,
What Is A Serial Dilution And Why Is The Technique Used
M1V1 = M2V2
In this problem, the initial molarity is 3.00 M, the initial volume is 2.50 mL or 2.50 x 10–3 L and the final volume is 0.175 L. Use these known values to calculate the final molarity, M2:
So, the final concentration in molarity of the solution is
4.29 x 10–2M
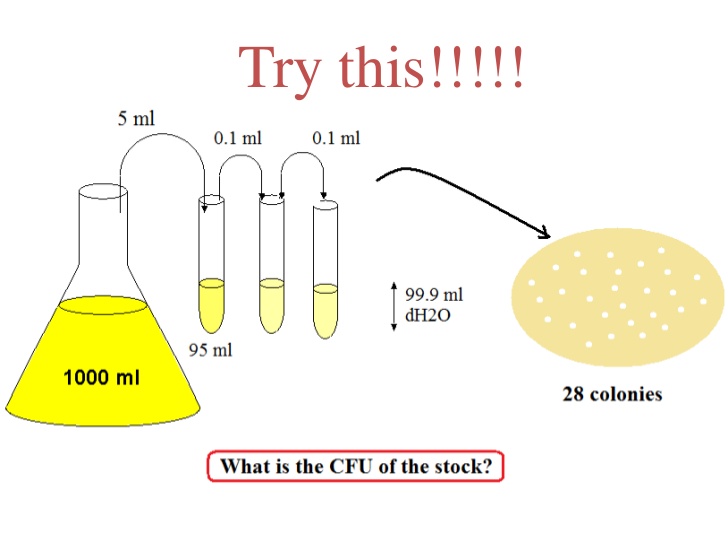
A serial dilution is the stepwise dilution of a substance in solution. Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion. A ten-fold serial dilution could be 1 M, 0.1 M, 0.01 M, 0.001 M .. Serial dilutions are used to accurately create highly diluted solutions as well as solutions for experiments resulting in concentration curves with a logarithmic scale. A tenfold dilution for each step is called a logarithmic dilution or log-dilution, a 3.16-fold (100.5-fold) dilution is called a half-logarithmic dilution or half-log dilution, and a 1.78-fold (100.25-fold) dilution is called a quarter-logarithmic dilution or quarter-log dilution. Serial dilutions are widely used in experimental sciences, including biochemistry, pharmacology, microbiology, and physics.
In biology and medicine[edit]
In biology and medicine, besides the more conventional uses described above, serial dilution may also be used to reduce the concentration of microscopic organisms or cells in a sample. As, for instance, the number and size of bacterial colonies that grow on an agar plate in a given time is concentration-dependent, and since many other diagnostic techniques involve physically counting the number of micro-organisms or cells on specials printed with grids (for comparing concentrations of two organisms or cell types in the sample) or wells of a given volume (for absolute concentrations), dilution can be useful for getting more manageable results.[1] Serial dilution is also a cheaper and simpler method for preparing cultures from a single cell than optical tweezers and micromanipulators.[2]
In homeopathy[edit]
Serial dilution is one of the core foundational practices of homeopathy, with 'succussion', or shaking, occurring between each dilution. In homeopathy, serial dilutions (called potentisation) are often taken so far that by the time the last dilution is completed, no molecules of the original substance are likely to remain.[3][4]
See also[edit]
References[edit]
How To Do Serial Dilutions
- ^K. R. Aneja. Experiments in Microbiology, Plant Pathology and Biotechnology. New Age Publishers, 2005, p. 69. ISBN81-224-1494-X
- ^Booth, C.; et al. (2006). Extremophiles. Methods in microbiology 35. Academic Press. p. 543. ISBN978-0-12-521536-7.
- ^Weissmann, Gerald (2006). 'Homeopathy: Holmes, Hogwarts, and the Prince of Wales'. The FASEB Journal. 20 (11): 1755–1758. doi:10.1096/fj.06-0901ufm. PMID16940145. Retrieved 2008-02-01.
- ^Ernst, Edzard (November 2005). 'Is homeopathy a clinically valuable approach?'. Trends in Pharmacological Sciences. 26 (11): 547–548. CiteSeerX10.1.1.385.5505. doi:10.1016/j.tips.2005.09.003. PMID16165225.
- Michael L. Bishop, Edward P. Fody, Larry E. Schoeff. Clinical Chemistry: Principles, Procedures, Correlations. Lippincott Williams & Wilkins, 2004, p. 24. ISBN0-7817-4611-6.
External links[edit]
What Is A 2 Fold Serial Dilution
- How to Make Simple Solutions and Dilutions, Bates College
Serial Dilution Calculator Microbiology
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Serial_dilution&oldid=904833671'
